In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the. 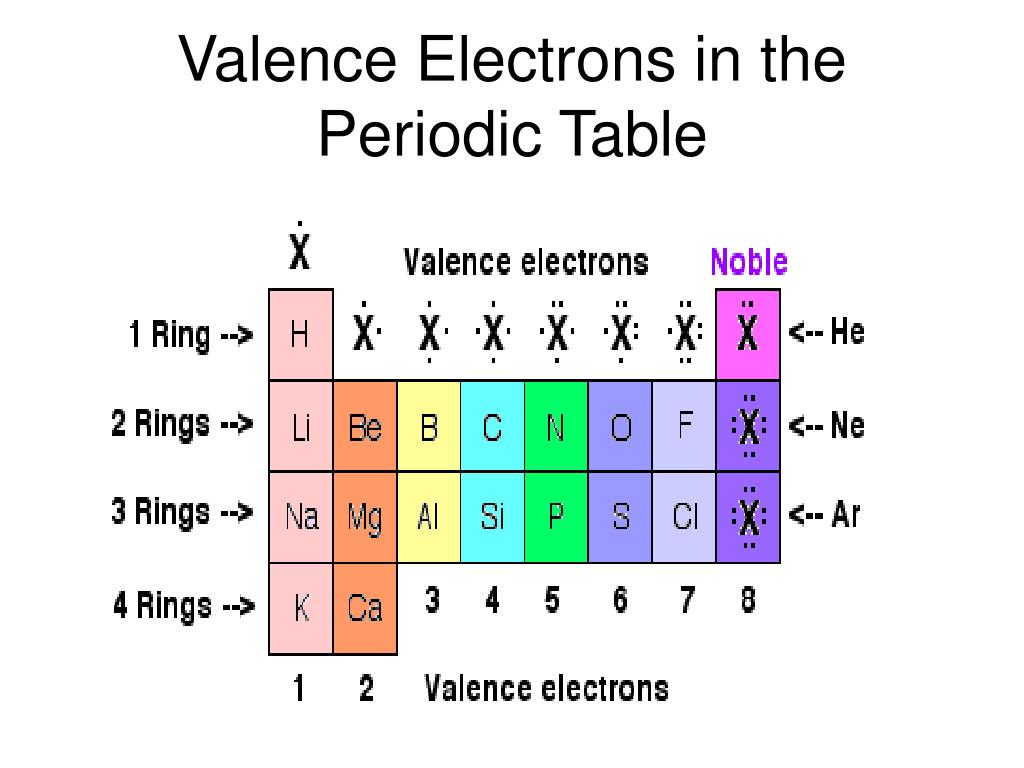
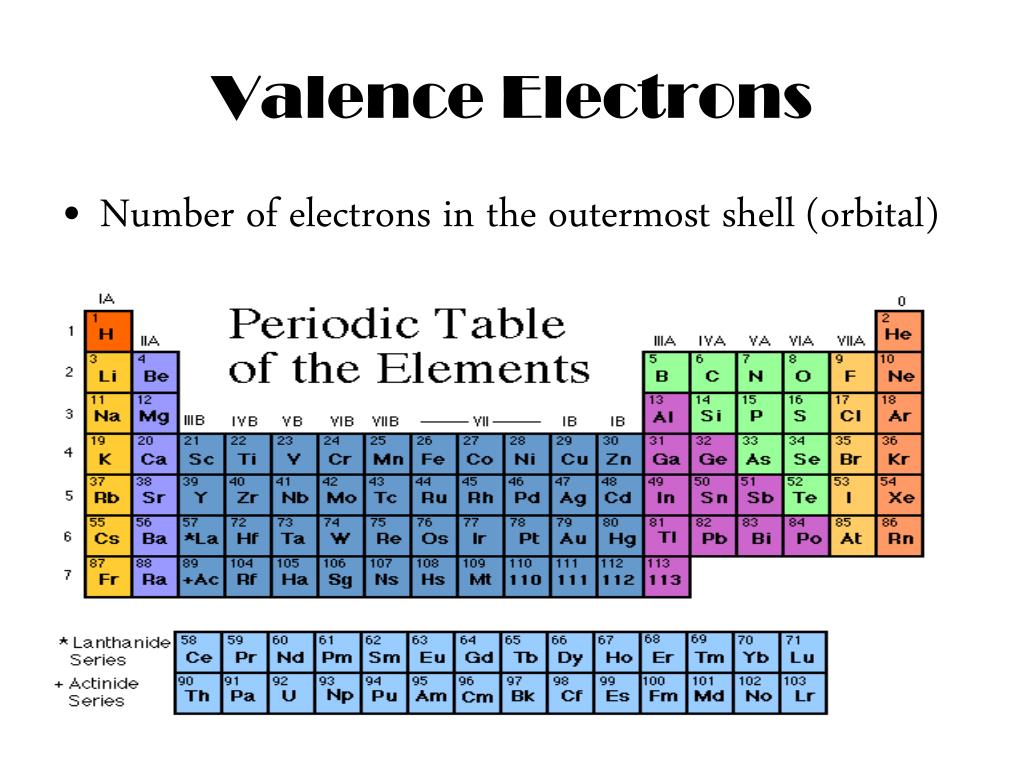
In other words, the number of valence electrons for a transition metal is equal to how many spaces across the periodic table it is. The group number (column number) represents the number of valence electrons common to all the elements within that group. As we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. The valence electrons for transition metals are equal to the number of s-electrons plus the number of d-electrons. For non-transition metals, we count to 8, but for transition metals, we count to 12. Looking at the orbitals explains how valence electrons work for transition metals. For example, oxygen has 6 valence electrons, these six electrons fill up the 2s orbital, and partially fill the 2p subshells (2s 22p 4). Electrons beyond the noble-gas configuration are valence electrons if they are in the outermost shell of the atom (have the highest n value) or if they are in. The shells after (ignoring transition metals) represent the s and p-orbitals. So what does this have to do with our shells? The first "shell" represents the 1s orbital. Since carbon has 6 electrons in total, it will have 4 valence electrons as shown below: Fig. All neutral elements will have the same number of protons and electrons, which is equal to the atomic number. For example, carbon has an electron configuration of 1s 22s 22p 2. Carbon is the 6th element in the periodic table. For instance, carbon is in Group 14, so it has four. The way we count our electrons is by moving from right to left, starting at the beginning of the table. For elements in Groups 13 to 18, the number of valence electrons is equal to the group number minus ten. F-orbitals start appearing in the lanthanides and actinides (the separated two rows). P-orbitals start appearing in period 2, and d-orbitals start appearing in period 4 (though they start counting at 3). Has 7 subshells, each holding 2 electrons, for a total of 14 electronsīelow is the periodic table with the labeled orbitalsĮach period is its own energy level.Has 5 subshells, each holding 2 electrons, for a total of 10 electrons.Has 3 subshells, each holding 2 electrons, for a total of 6 electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
